Clinical trials are hard to do and often requires the cooperation of the CRO to execute the study, but Also, the study center that is recruiting The participants in the clinical study. The CRO monitors the clinical study but also all the aspects of the clinical study.
A company called T3D therapeutics (https://www.t3dtherapeutics.com/) recently had a study not done correctly due to the recruitment that was not executed well by their CRO provider called Clinilabs (https://clinilabs.com/) who recruited participants from suspect medical centers and made their study iirrelevant.
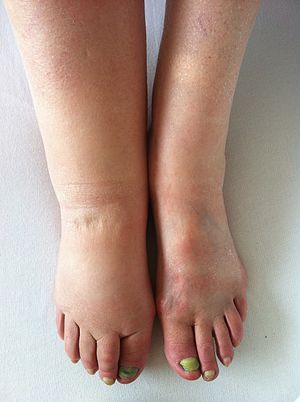
This was written up in science (https://www.science.org/content/article/alzheimer-s-drug-developers-accuse-clinical-trial-sites-faking-data) and also by Derek Lowe (https://www.science.org/content/blog-post/clinical-trial-nightmare) which said that company received medically impossible data from placebo participants. This seems to be specific for some South Florida clinical trial sites but wonder how did Clinilabs recruit these sites and participants?