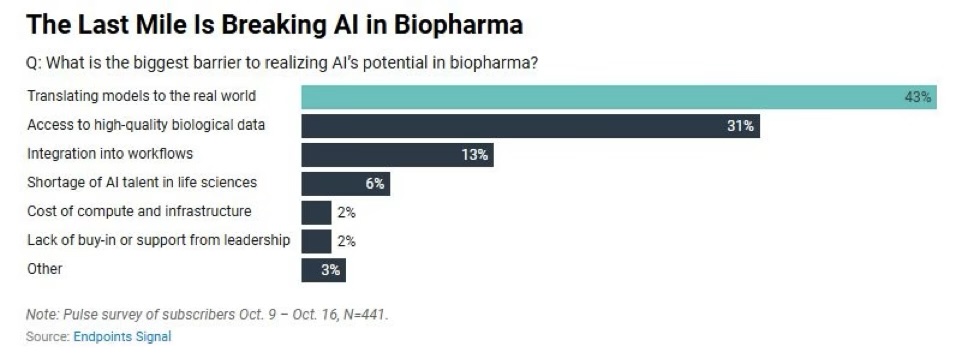
This was in a LinkedIn Post by Jeffrey Low and was from a survey done by Endpoints news and it is so correct that it is very difficult to translate animal models to human biology. This is coming from a group of about 500 verified subscribers who were asked in October 2025. The survey also pointed out that many of the believers in AI were the senior executives, though the business development folks did not believe it would make a big difference. The article ended with a comment from the CTO at Amgen – David Reese – who commented that it will not be a big bang for AI in Biopharma but rather a continuous adoption of process that will optimize many parts.
Lets think about the the top issue:
Translational gap. Why is it difficult to translate in Biotech and Pharma? The fact that it estimated that nearly 90% of drugs that go through human testing fail due to lack of efficacy in the human clinical study. There are many reasons. Lets outline a few. A key fact is that discoveries are made in a simplified, controlled system (whether that is a at a cell level or in a mouse living in a germ free environment) whereas humans are genetically variable living with a complex multi-organ physiology in a diverse environment. There are many other reasons – species and system differences, practical considerations for doing such studies, ethical considerations for human studies. The examples are many…for example p53 works as a perfect tumor suppressor but the human results are more nuanced, the p53 mutations are only partially inactivated. Or consider anti-TNF therapies – they work well in mouse arthritis models but are more variable in humans.
Is there a solution? There have been many proposed, such as doing more human studies and forming consortium. The most promising may be the compilation of human data to use for future studies. This had been difficult in the past since the transcriptomics, proteomics and the multi-omic data but new AI techniques may make that possible.

